Is H2o Molecule Polar or Nonpolar
In such a bond there is a charge separation with one atom being slightly more positive and the other more negative ie the bond will produce a dipole moment. Covalent bonds in which the sharing of the electron pair is unequal with the electrons spending more time around the more nonmetallic atom are called polar covalent bonds.
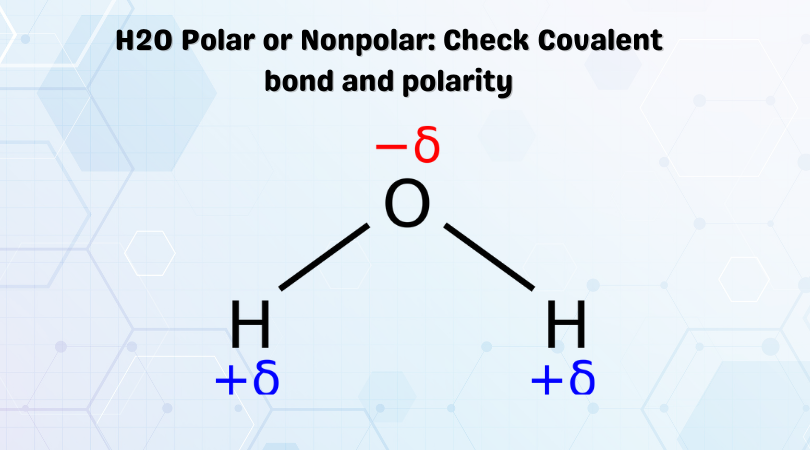
H2o Polar Or Nonpolar Check Covalent Bond And Polarity Geometry Of Molecules
Mathematically it is the product of the charge on atoms and the distance between them.
. The polar molecule always has a non zero dipole moment. And the nonpolar molecules have zero dipole moment. The shape of a molecule is an important parameter to check whether a molecule is polar or not.
9 6 Polar And Non Polar Molecules Physical Science
Similar H2o Consists Of Polar Molecules Co2 Consists Of Nonpolar Molecules How Do Chemists Explain This Difference Quora

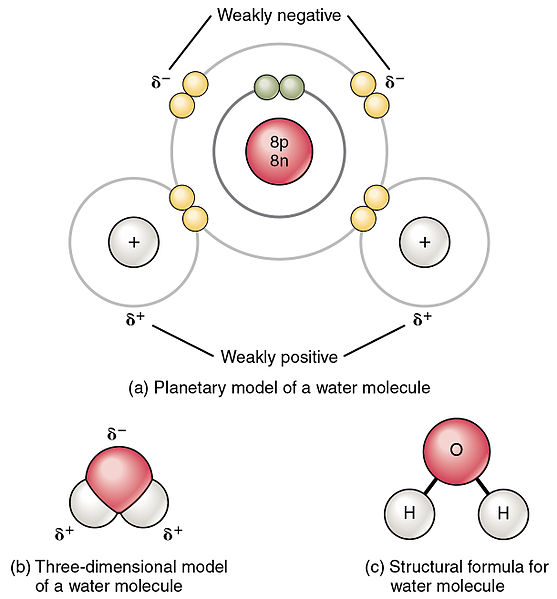
Covalent Bonds Biology For Majors I
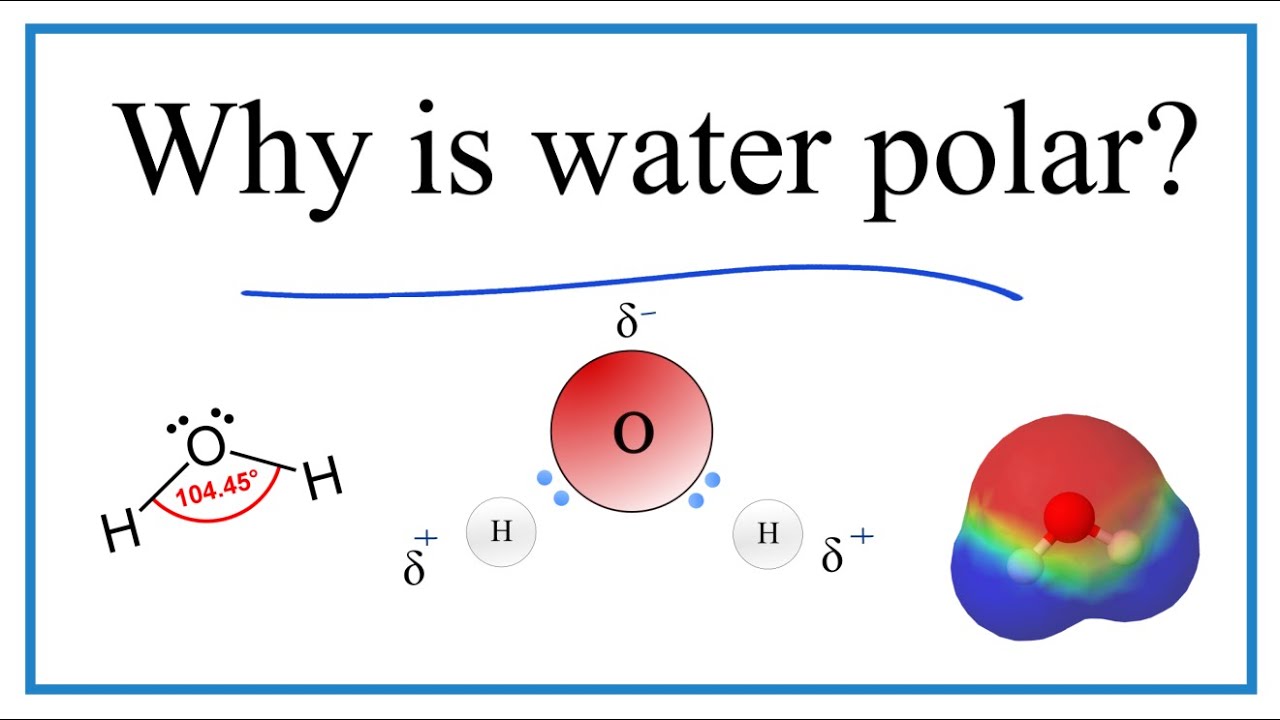
Why Is Water H2o A Polar Molecule Youtube
Is The Molecule H2o Polar Or Non Polar Quora

Polar And Nonpolar Covalent Bonds Definitions Molecules And Examples

Are Hydrophilic Molecules Polar Or Non Polar Quora

Is H2o Water Polar Or Nonpolar Techiescientist

Types Of Covalent Bonds Polar And Nonpolar Manoa Hawaii Edu Exploringourfluidearth

Polar And Nonpolar Covalent Bonds Characteristics Differences

Is H2o Water Polar Or Nonpolar Techiescientist

Chemistry Ii Water And Organic Molecules

Is H2o Polar Or Nonpolar In Urdu Hindi Covalent Bond Chemicalbonding Youtube Youtube
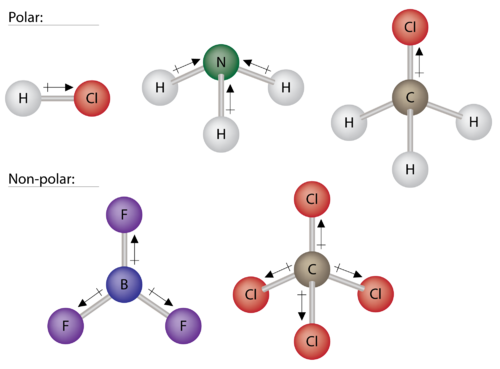
Polar Molecules Chemistry For Non Majors

Polar And Non Polar Molecules Tmjh 8th Grade Science


Comments
Post a Comment